
Factors to consider in making scleral lens fits as successful as soft lens fits for normal corneas.
A new way of clinical thinking is needed when it comes to fitting scleral lenses on normal corneas. Traditionally, scleral lenses are considered for diseased eyes or to compensate for highly irregular corneal surfaces. The positive outcomes from their use in such patients are numerous, and in such cases it is easy to determine that the benefit/risk ratio is high (van der Worp et al, 2014).
However, on a normal cornea, does this still hold true? Using scleral lenses to correct the regular refractive error of a 20-year-old patient or a 15-year-old baseball player now makes sense in theory. The benefits potentially include excellent vision and reduced contact lens-induced discomfort. However, we know very little about the physiological impact of scleral lens technology over the long term. Is the benefit/risk ratio still considered high for these patients?
Benefits of Scleral Lenses for Normal Corneas
The first factor to consider is that scleral lenses can resolve the two major reasons why current soft contact lens wearers drop out of lens wear: discomfort and visual acuity issues; the latter is typically more prevalent in astigmatic and presbyopic patients (Rumpakis, 2010).
Contact lens-induced discomfort affects up to 50% of soft lens wearers (Riley et al, 2006) and, despite improvements in lens materials and care regimens, still represents a day-to-day challenge for patients and practitioners. Scleral lenses are filled with fluid to keep the ocular surface constantly bathed. Consequently, they are less associated with factors related to lens-induced discomfort compared to soft lenses, so they can help normal cornea patients who experience lens-induced dryness (Alipour et al, 2012).
Visual acuity is improved with the use of GP lenses in general; with sclerals, it is even better. This is true because these lenses offer a larger optical zone compared to any other lens type. Most soft lenses have a 6mm to 7mm optical zone diameter, which is reduced to 4mm to 5mm with toric designs. Most scleral lenses offer an 8mm to 9mm optical zone diameter. Even small GP lenses cannot compete with sclerals in that regard. As a result, scleral lenses reduce halos and glare, with a corresponding improvement in vision, even under the most challenging conditions.
Another source of visual pollution is high-order aberrations (HOAs), which tend to occur in patients who have irregular corneas or lenticular opacities (Maeda, 2002). Ophthalmic lenses also induce a certain amount of HOAs, mostly spherical aberrations. In soft lenses, a custom design can compensate for some HOAs, but not for all. The tear fluid under a scleral lens helps to reduce HOAs by eliminating corneal surface irregularities.
People fitted in soft toric contact lenses could experience fluctuating vision while biking or running in windy conditions. This is not the case with scleral lenses, primarily because they remain stable on the eye. Athletes are often exposed to challenging environments. Sand, wind, and dust can create discomfort in people who wear soft lenses and, in particular, those fitted with small GP lenses. Scleral lenses provide a sealed ocular surface environment. Once the lens has settled, no particles can touch the cornea or invade the space between the lens and the ocular surface. Scleral lenses can even protect against trauma (Altman, 2012; Walker, 2015).
When all of these elements are considered, it makes sense to adopt scleral lens technology for the correction of more regular refractive errors and as a contact lens modality of choice for patients who engage in sports activities, particularly in challenging environments.
What About Oxygen Delivery to the Cornea?
It was suggested some years ago on the basis of a theoretical model that oxygen delivery to the cornea would be impacted if the scleral lens was 350 microns thick and the tear fluid thickness (clearance) was to exceed 200 microns (Michaud et al, 2012). This theoretical model has been proven valid clinically with signs of induced hypoxia after several hours of wear when a scleral fit exceeds those parameters (Compañ et al, 2014). More recently, a new theoretical model added to this theory, suggesting that corneal oxygen consumption would be higher after a patient has been wearing thick scleral lenses fitted with a higher clearance (Jaynes et al, 2015).
Today it is acknowledged that large scleral lens wear is associated with chronic oxygen deprivation, leading to 2% to 3% of edema at the end of the wearing period if the lens is thick (more than 300 microns) and the clearance remains high over the wearing period (more than 250 microns) (van der Worp et al, 2014). It is also suggested that this level of edema is not clinically significant, being very similar to the level of physiological edema present upon awakening (Caroline et al, 2015).
However, it is misleading to suggest such a comparison because in the latter instance, oxygen restoration occurs within the first 60 minutes after eye opening, and the cornea remains free of edema for the rest of the day. In large scleral lens wear, edema remains present throughout the wearing period.
At best, there is a transient hypoxic stress applied to the cornea. The clearance is high in the first few hours of wear, but it will be reduced as the lens settles on the conjunctiva.
According to the diffusion models, the initial higher clearance will generate hypoxia, but the tissue will be able to recover as soon as lens settling reduces the clearance, during the last hours of wear. This is probably why, clinically, on a healthy cornea, it is difficult to identify signs of oxygen deprivation with large scleral lens wear. In addition, edema becomes visible at 4% and greater. If it remains overall at 2% to 3%, it is conceivable that it may not be observed during a slit lamp examination.
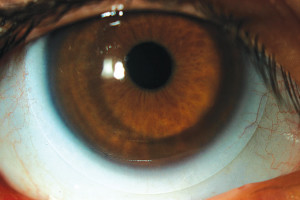
Figure 1. A smaller scleral lens on a normal cornea.
Visible or not, the real issue is that, at this point, nobody knows the effect of exposing a normal cornea to chronic 2% to 3% central edema over a long time period. For patients who have irregular corneas or diseased eyes, the risk is minimal compared to the important benefits of restoring visual acuity or treating the ocular surface. However, on a non-compromised, normal cornea, or on a post-graft patient who has a compromised endothelium, the presence of chronic edema significantly decreases the benefit/risk ratio.
Think about a healthy 15-year-old baseball player looking to wear contact lenses for 10 to 20 years, or even longer. Would it be ethical to suggest a modality associated with chronic central corneal edema, with no information as to the long-term effects of such a condition?
Consider Smaller Sclerals for Normal Corneas
The only safe way to proceed in such cases is to fit lenses that can meet the cornea’s oxygen delivery requirements from application to removal. Smaller scleral lenses (14mm to 15mm) are manufactured thinner (250 to 300 microns) and do not need higher clearance to support them. When fitted on regular corneas (Figure 1), these lenses do not need high clearance to vault over this predictable and smooth surface (Figure 2). Consequently, smaller sclerals can be worn without inducing chronic edema on the corneal tissue at any time. These lenses have a very high benefit/risk ratio compared to the larger scleral lenses.
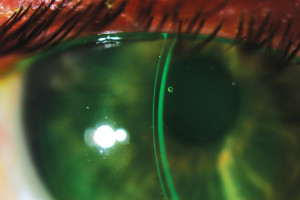
Figure 2. Optimal tear layer thickness and lens thickness for a smaller scleral lens.
The process of fitting smaller scleral lenses is relatively similar to that required with larger ones. Both types of scleral lenses aim to vault over the cornea without touching it. They also vault over the limbus, even though the clearance is always reduced over this area (Figure 3). With smaller lenses, this reduced fluid layer (around 40 microns) over the limbal area helps increase oxygenation to this sensitive tissue, and it facilitates the production of a wedge-shaped tear reservoir, which helps support a significant portion of the lens weight.
This element is important to understand because some practitioners believe that there is not enough diameter with smaller scleral lenses to be well supported on the sclera. They may not realize that the fluid reservoir is not only beneficial for refractive purposes, it also contributes mechanically to the scleral lens behavior on the eye, supporting its weight and acting like a water bed.
Limited clearance has also been identified as a factor that could have an impact on stem cells (DeNaeyer, 2015). There are two types of potential impact: physiological and mechanical. Physiologically speaking, some stem cells need more oxygen compared to the central cornea; some prefer some level of hypoxia (Bath et al, 2013). Limiting the clearance over stem cells serves to increase oxygen delivery. Higher clearance over the limbus can indeed have a more negative impact on stem cells compared to the converse.
Mechanically speaking, stem cells are located deep in the corneal tissue, and it is difficult to believe that a limited clearance would challenge them as long as the weight of the lens is well distributed across the entire limbal surface. In fact, corneo-scleral lenses fitted in this way have never been associated with neovascularization or stem cell deficiencies.
Finally, limited clearance over the limbus is associated with a reduced risk of developing tear reservoir fogging over the wearing period (Caroline, 2014). A higher clearance over the limbus allows lipids and mucin to diffuse into the tear reservoir. The fluid becomes milky in appearance and impacts visual acuity. In such cases, patients need to remove the lens, rinse it, and replenish its fluid content. Having to remove the lens to replenish the tear reservoir is not convenient for any patient. Patients who have diseased eyes or irregular corneas will do it if they have to, because their other options are limited. However, patients who have normal corneas will not continue wearing scleral lenses if doing so becomes less convenient compared to other modalities.
Conversely, with smaller sclerals, which are associated with limited limbal clearance, lipids and mucins cannot migrate under the lens, and the fluid reservoir remains free of debris during the entire wearing period. Consequently, the purported association between limited limbal clearance and negative clinical issues is a myth that we have to identify and dispel if we want to fit normal corneas with scleral lenses.
Another difference with smaller scleral lenses is that there is usually no need to modify their peripheries to optimize their fit. It is now established that scleral toricity must be considered when lenses exceed 15mm in diameter (van der Worp, 2014). Many manufacturers now offer the option of toric haptics to manage scleral toricity; however, it is a complicated process. With smaller scleral lenses, the lens is landing on a part of the eye that is usually not toric in nature. Therefore, these lenses do not typically have to be optimized with toric haptics.
Handling is easier with smaller scleral lenses. They are roughly the same size as soft toric contact lenses and are easy to apply and remove using orthodontic rubber bands or just the fingers, similar to soft lenses. For practitioners, this makes the application and removal training process easier; for patients, it is less intimidating. Ease of handling is an important factor when considering scleral lenses for normal corneas. We have to differentiate between patients who have irregular corneas and/or diseased eyes and those who have normal corneas. In the first instance, patients will become accustomed to the larger diameter and will learn to handle the lenses because there are not really any other options for them. However, this is not the case for patients who have normal corneas and for whom soft lenses are still a possibility.
Realistically speaking, if practitioners and manufacturers want to promote sclerals as mainstream products for the correction of normal refractive errors, they need to minimize the negative aspects for patients. Handling issues still represent the number one reason for failure in refitting these patients (personal clinical experience; Schornack and Patel, 2010). Convenient designs and appropriate training are the answers to this.
The Case for Presbyopia
Modern mini-scleral and scleral lenses are becoming available in presbyopic designs. By vaulting over the cornea, spherical scleral lenses can compensate for high levels of corneal astigmatism without the need to rely on a front-toric design, making them suitable for most presbyopic patients. The second important factor to consider for presbyopes is that their tear film is more unstable compared to younger lens wearers (Sweeney et al, 2013). Alongside other treatments, scleral lenses can help these patients by providing relief from ocular dryness.
Optically speaking, scleral lenses are an ideal way to correct presbyopia. They offer a larger optical zone than any other contact lens, and they do not move on the eye. Centration is crucial to obtain a good outcome in multifocal contact lens fitting. This is even more true with scleral lenses, and the diameter of the lens is a factor in this regard. Larger scleral lenses tend to decenter inferiorly and temporally (DeNaeyer, 2015), and therefore the optical axis of the lens often does not correspond to the visual axis of the patient, which impacts visual performance. In that regard, smaller scleral lenses, which are less inclined to decenter, are a better option for presbyopic patients.
A Case in Point
Jane is a 46-year-old computer graphics instructor who requested a consultation to address vision problems that she has been experiencing while working with her students at the computer. She wears soft toric two-week replacement contact lenses and would like to keep them as a modality of choice to correct her vision. She reports no major issues with her current lenses, except for a certain amount of dryness by the end of the day. She also reports fluctuating vision while biking or training outside.
What options are available for refitting this patient? Unfortunately, not many. Reading glasses over monofocal contact lenses are not an option because the patient does not want to look too old too soon. Soft toric multifocal frequent replacement contact lenses do exist, but they can be associated with reduced contrast sensitivity and poorer results under reduced illumination (Madrid-Costa et al, 2012).
Masking astigmatism with spherical lenses is not recommended, especially for presbyopic patients. Over-minusing an eye leads to increased accommodative and convergence demands, which is not the best strategy for a presbyope or for a patient working at computer distance. Uncorrected astigmatism reduces the clarity and sharpness of an image, increases blur at intermediate distance, and is associated with higher ocular fatigue. In addition, many studies have established the value of correcting a small amount of refractive astigmatism (0.75D or more) for all patients (Cho et al, 2012; Richdale et al, 2007).
Monovision could be an option, but it is increasingly less prescribed because of the proven value of maintaining full binocular vision, especially for presbyopes (Richdale et al, 2006). In this particular case, monovision was excluded because the patient needed good depth perception for both her work and her sports activities.
GP contact lenses are the best way to correct the combination of myopia, astigmatism, and presbyopia. They provide crisp and sharp vision at all distances and offer many different designs to address patients’ visual needs. However, small-diameter GP lenses are associated with initial and long-term discomfort, and more importantly, multifocal aspheric designs may induce corneal warpage over time. Hybrid contact lenses can help by improving the rigid lens centration, and they are not typically associated with corneal distortion. However, they may not be able to resolve the patient’s dryness symptoms.
What other options remain? I believe that smaller scleral lenses may be the best option for this patient, considering her symptoms of induced dryness (Messer et al, 2015).
Troubleshooting Issues with Smaller Scleral Lenses
Some complications with smaller scleral lenses are the same as with larger scleral lenses. Discomfort 20 to 30 minutes post-application is linked to the presence of air bubbles in the reservoir. Using a more viscous product to fill the lens for application can help patients who experience chronic bubble issues. However, more viscous filling solutions can impact both vision and oxygen diffusion.
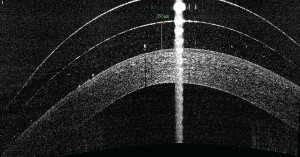
Figure 4. An optical coherence tomography image showing too much clearance for a mini-scleral lens.
Discomfort after four to six hours of wear is associated with tight lenses. They compress the conjunctival vessels and seal off the ocular environment. Reduced central clearance or flatter peripheries will help alleviate this problem.
Smaller scleral lenses that move on the surface of the eye are fitted with too much clearance (Figure 4), which has to be reduced to improve lens behavior.
Residual astigmatism can also be an issue. This comes from two sources. Because the fluid reservoir compensates for anterior corneal astigmatism, posterior corneal or lenticular astigmatism can become visible once the lens is fitted. This can be corrected with a front-toric scleral lens design, which requires a slightly larger diameter (15mm to 16mm) to stabilize the correction on the ocular surface.
It has also been thought that residual astigmatism could result from lens flexure on the eye. This is a second myth that needs to be dispelled. As has been shown through keratometry, this results more from a torsion of a spherical lens on a toric surface than from true flexure of a thick lens. The proof comes from the fact that increasing the lens thickness results in almost the same amount of residual astigmatism. Not only will increased scleral lens thickness penalize oxygen delivery, it will also increase the lens weight and its tendency to decenter. In fact, the irregular shape of the tear reservoir is the primary reason behind most of the residual astigmatism with scleral lens wear. It is therefore possible to address this by increasing the clearance (if this is possible without penalizing oxygen delivery) and/or by increasing the optical zone diameter and modifying the midperipheral curves to make the reservoir more uniform in shape.
Conclusion
Today, there is general consensus that mini-scleral lenses (14.5mm to 15.5mm in diameter) are easier to fit and, for patients, as easy to handle as soft lenses. Smaller scleral lenses are ideal for correcting regular refractive errors, including astigmatism and presbyopia, as well as a low-to-moderate level of corneal irregularity. Larger lenses (15.8mm to 18mm in diameter) are more commonly used for managing severe eye dryness (Stevens-Johnson syndrome, Sjögren’s syndrome) and for greater corneal irregularity (pellucid marginal degeneration, for example). Larger sclerals are also ideal if a front-toric design is needed, because they are more easily stabilized.
In general, practitioners should rely on more than one trial set to be able to fit a normal cornea patient with scleral lenses. This also takes a new way of thinking clinically. For specific fitting tips, it is important to consult with the lens manufacturers and to become educated and trained appropriately. The Scleral Lens Education Society website (www.sclerallens.org) is a good place to start to learn about scleral lens technology. Well-fitted mini-scleral or scleral lenses will help reduce end-of-day discomfort, glare, halos, and fluctuating vision issues. In other words, they can help practitioners keep their contact lens patients in the market and reduce the dropout rate.


